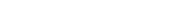Are Cold Packs Exothermic Or Endothermic? Exploring Temperature Changes
TÓM TẮT NỘI DUNG
Hot Pack Vs Cold Pack – Exothermic Vs Endothermic Chemical Reactions
Keywords searched by users: Are cold packs exothermic or endothermic why is a cold pack endothermic, is a hot pack endothermic or exothermic, are hand warmers endothermic or exothermic, is melting endothermic or exothermic, is ice melting endothermic or exothermic, why is ammonium chloride used in cold packs, is endothermic hot or cold, is a burning match endothermic or exothermic
Why Are Cold Packs Exothermic?
“Why do cold packs generate heat? To answer this question, let’s delve into the inner workings of these ingenious devices. Within a cold pack, you’ll find a critical component: a metal clicker. When the cold pack returns to room temperature, it undergoes a fascinating transformation. This transformation involves the crystallization of sodium acetate, which is an exothermic process, meaning it releases heat. The metal clicker plays a crucial role in this process by providing a nucleation point for the sodium acetate to crystallize around. Cold packs are typically composed of ammonium salts, as indicated by their stated contents, and it’s this combination of substances and the exothermic reaction of sodium acetate crystallization that makes cold packs exothermic, producing the comforting warmth you feel when using them.”
Is A Hot Pack Exothermic Or Endothermic?
Is a hot pack exothermic or endothermic? Understanding the thermodynamics behind hot packs helps clarify this. The operation of a hot pack involves an energy exchange with its surroundings. Initially, energy is absorbed from the surroundings to break the bonds within the pack’s contents, often involving ionic compounds. These ions subsequently establish new bonds with water molecules, and this bond formation releases energy. When the energy released during this process exceeds the energy initially absorbed, the overall reaction is deemed exothermic. Consequently, the hot pack feels warm to the touch as a result of this exothermic process. In contrast, if more energy is consumed than released, it leads to an endothermic reaction, making the pack feel cooler. This interplay of energy transfer explains whether a hot pack is exothermic or endothermic.
Is A Chemical Cold Pack An Example Of An Exothermic Process?
Is a chemical cold pack considered an instance of an exothermic process? To answer this question, let’s explore the science behind it. A chemical cold pack cools down due to an endothermic reaction. Inside the pack, there’s a sealed tube containing ammonium nitrate, a chemical that reacts with the water contained in the pack. This reaction is endothermic, meaning it absorbs heat energy from its surroundings. Consequently, the contents of the pack rapidly cool down. It’s important to note that while this particular chemical process is endothermic, many other chemical reactions can be exothermic, releasing heat energy instead of absorbing it.
Update 40 Are cold packs exothermic or endothermic






Categories: Update 33 Are Cold Packs Exothermic Or Endothermic
See more here: cuahangbakingsoda.com

An instant cold pack is the perfect example of an endothermic reaction. There are many possible ingredients in an instant cold pack, but they often contain solid ammonium nitrate and water. Did you know? Ammonium nitrate is a nitrate salt.Inside is a metal clicker, once the pack cools to room temperature, crystallization of sodium acetate (an exothermic process) is triggered by the clicker, which gives the sodium acetate a nucleation point. Cold packs are presumably ammonium salts (judging by the stated contents).This process requires energy, which is obtained from the surroundings. The ions then form bonds with the water, a process that releases energy. If more energy is released than taken in, then the process is exothermic, making the solution feel warmer.
Learn more about the topic Are cold packs exothermic or endothermic.
- The Cold Pack: A Chilly Example of an Endothermic Reaction
- T520: Exothermic and Endothermic Reactions – Hot and Cold Packs
- Hot and Cold Packs: A Thermochemistry Activity
- 3.9 Energy in Chemical Reactions – Human Biology
- How Do Instant Ice Packs Work? – Steroplast Healthcare
- What’s Inside an Ice Pack? | Poison Control