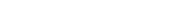Are Diamond And Graphite Allotropes? Exploring Their Unique Structures
TÓM TẮT NỘI DUNG
Gcse Chemistry – Allotropes Of Carbon – Diamond And Graphite #18
Keywords searched by users: Are diamond and graphite allotropes diamond and graphite are allotropes of carbon what does that mean, diamond and graphite are of carbon, diamond allotropes of carbon, why is diamond so hard, diamond is an allotropic form of, allotropes of carbon diagram, allotropes of carbon pdf, name the allotropes of carbon
Is Graphite An Allotrope?
Graphite is indeed a noteworthy allotrope of carbon, representing one of its crystalline forms. This versatile substance exists in both natural and synthetic variants. Notably, graphite exhibits remarkable thermal and electrical conductivity properties, making it an essential material with a wide array of applications in various industries.
Are Diamond And Graphite Allotropes Or Isomorphous?
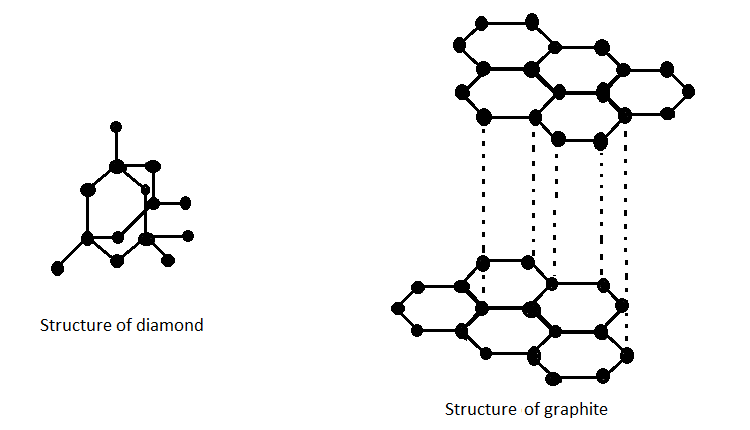
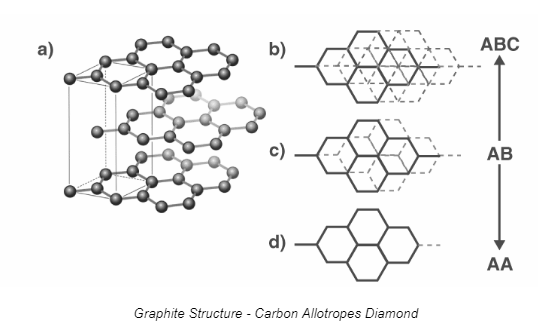
Diamond and graphite are two distinct structural forms of carbon, known as allotropes. Allotropes are different arrangements of atoms of the same element in various crystalline structures. In the case of carbon, these two allotropes exhibit vastly different properties due to their unique atomic arrangements. Diamond consists of carbon atoms arranged in a tetrahedral lattice, making it one of the hardest natural materials with exceptional transparency and brilliance. In contrast, graphite is composed of layers of hexagonally arranged carbon atoms bonded together in sheets, imparting properties such as lubricity and electrical conductivity. Therefore, while both diamond and graphite are allotropes of carbon, they display stark differences in their physical and chemical characteristics due to their distinct atomic structures.
Collect 28 Are diamond and graphite allotropes



Categories: Top 64 Are Diamond And Graphite Allotropes
See more here: cuahangbakingsoda.com

Diamond and Graphite are allotropes of carbon. They consist of carbon atom atoms but they have different physical properties. They are polymorphs having the same chemistry but the crystalline structures are different.Graphite is an important allotrope carbon and is a crystalline allotrope form. Graphite is available in both natural and synthetic forms. Graphite is an excellent conductor of heat, and electricity has a range of uses of graphite.Diamond and graphite are allotropes of carbon. Reason: Some elements can have different structural forms called allotropes.
Learn more about the topic Are diamond and graphite allotropes.
- Why do diamonds and graphite, the two allotropic forms of …
- Allotropes of Carbon – Uses of Graphite, chemistry by unacademy
- How graphite and diamond are isomorphous when they … – BYJU’S
- Allotropes of carbon | Feature – RSC Education
- Diamond and Graphite – Structure, Uses, Properties, Applications
- [Solved] Which of the following is NOT an allotrope of carbon? – Testbook