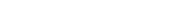Are Electrons Always In Motion? Exploring The Constant Dance Of Subatomic Particles
TÓM TẮT NỘI DUNG
Where Do Electrons Get Energy To Spin Around An Atom’S Nucleus?
Keywords searched by users: Are electrons are constantly moving moving electrons are called, electrons are in constant motion around the nucleus because of, can protons move, electron move from positive to negative, can a neutron move, electrons that spin in, can electrons stop moving, electrons move around the nucleus
Are Electrons Always Moving?
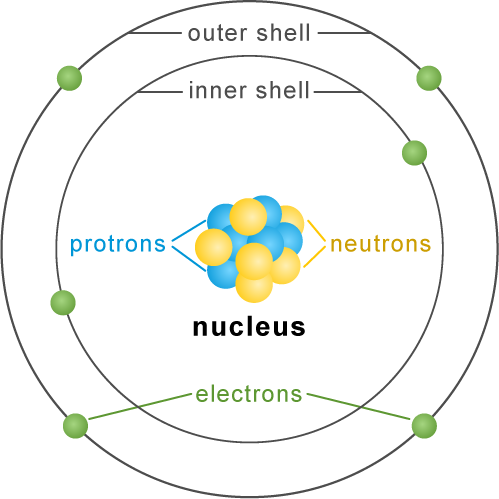
Do electrons always remain in motion? To answer this question, it’s essential to understand the behavior of electrons within atoms. In general, electrons within atoms do not exhibit classical motion, as they don’t follow predictable orbits like planets around the sun. Instead, electrons in their lowest energy state, as seen in the hydrogen atom, are primarily characterized by their stationary electron cloud. This electron cloud maintains a fixed distribution over time, meaning that the electrons do not travel in the conventional sense. However, it’s crucial to note that these electrons still possess kinetic energy, which is associated with their radial velocities within the electron cloud, contributing to their overall dynamic nature.
Are Electrons Always In Constant Motion?
Do electrons always remain in a state of perpetual motion within the electron cloud? Indeed, electrons are in a state of constant movement, a phenomenon that poses a unique challenge to scientists attempting to determine their precise whereabouts. This perpetual motion within the electron cloud renders it difficult for researchers to pinpoint the exact location of electrons within an atom.
Top 28 Are electrons are constantly moving



Categories: Details 36 Are Electrons Are Constantly Moving
See more here: cuahangbakingsoda.com

Moreover, the motion of these particles is not continuous but discontinuous and random in nature. We may say that an electron is a quantum particle in the sense that its motion is not continuous motion described by classical mechanics, but random discontinuous motion described by quantum mechanics.1. Generally atoms’ electrons do not move in anything like the classical sense. In particular, for the lowest energy state of the hydrogen atom, the electron cloud goes absolutely nowhere, keeping a fixed distribution in time. There’s some kinetic energy, associated with a distribution of purely radial velocities.Answer and Explanation:
Electrons are constantly in motion in the electron cloud. These electrons are constantly in motion, and therefore scientists are unable to pinpoint the exact location of electrons.
Learn more about the topic Are electrons are constantly moving.
- How do electrons move in atoms? From the Bohr model to …
- How do Electrons Move in Atoms? | Physics Van | UIUC
- Where are electrons in constant motion? – Homework.Study.com
- Voltage – Nondestructive Evaluation Physics : Electricity
- This is not what an atom really looks like – Big Think
- Why don’t electrons in an atom stop moving? – Physics Stack Exchange